Extended resection of non-small cell lung cancer invading the left atrium, is it worth the risk?
Introduction
Surgical resection remains a critical component of multidisciplinary therapy for locally advanced non-small cell lung cancer (NSCLC), thus, extended resection may improve survival of selected patients. Left-atrial resection is infrequently performed and surgical techniques are rarely reported; moreover, oncological results and survival rates remain uncertain. Currently, 5-year survival rate is about 24% in stage IIIA and 9% in stage IIIB (T4N2M0) disease (1). However, with modern advances in imaging, surgical techniques and perioperative care, highly selected patients may benefit from multimodal therapy.
Surgical procedure
With the advent of cardiopulmonary bypass (CPB) in the late 1950s, thoracic surgeons realized the potential for extracorporeal circulation to facilitate resection of pulmonary malignancies invading the heart or great vessels. Bailey and colleagues (2) were the first to report an extended left pneumonectomy invading the left atrium performed under CPB. Two patients had been operated on: the first one died in the early postoperative course from coagulopathy, whereas, the 2nd patient survived 14 months before succumbing to metastatic disease.
The use of CPB in cancer surgery has been debated for many years: it has been thought to increase the risk of disseminated metastasis or the likelihood of recurrence. Only recently, Muralidaran and colleagues (3) demonstrated that planned CPB usage does not decrease patients’ life expectancies. It’s the surgeon’s duty to estimate how much of the left atrium has to be removed to ensure en bloc R0 resection. Based on preoperative CT-scan and cardiac MRI, the thoracic surgeon must evaluate if a patch graft will be necessary in order to maintain a functional left atrium, if so, CPB is mandatory. Experimental studies on dogs (4) showed that up to a third of the left atrium could be resected without hemodynamic disturbance. However, to our knowledge, no study in humans has provided precise information on this limit.
We know that right-sided tumors can invade the left atrium faster than left-sided tumors, as pulmonary veins are shorter. This unfavorable anatomical condition is counterbalanced by good exposure of the right aspect of the left atrium, the ending of the right pulmonary veins, and the junction of the two atria. Moreover, this junction is superficially marked by the interatrial groove, and can be dissected to increase the length of the atrial cuff.
In our institution, we have developed a surgical procedure based on the dissection of the interatrial septum (IAS) in order to increase surgical margins without the need of CPB in case of right-sided pulmonary resection extended to the left atrium. Our surgical and oncological results have been published recently (5).
Surgical technique
Under general anesthesia and selective bronchial intubation, a muscle-sparing lateral thoracotomy or a postero-lateral thoracotomy is performed. Resectability is ensured prior any further step. The dissection begins in the sulcus, just behind the connection to the superior vena cava with the right atrium, and is continued caudally where the sulcus is hardly noticeable. The atrial roof attaching the superior and anterior aspects of both atria is composed of interatrial muscles fibers. This musculature leads to the posterior aspect of the aortic root and increases the length of dissection in the upper part of the interatrial groove by two-fold (facing the upper pulmonary vein). The limits of depth of the dissection are represented by the limbus of the fossa ovalis and by the Koch triangle at the lower extremity. In addition, the resectable left atrial cuff can be a mean distance of 40 mm at the level of the upper pulmonary vein after sectioning the interatrial muscle (Figure 1).
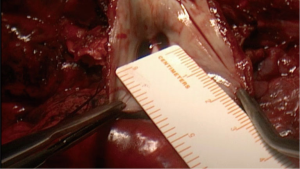
We propose 3 levels of left-atrial resection (5) in order to precise the operative technique used. The IAS can be dissected down to the ascending aorta root. The extra-length of left-atrial cuff permits a complete resection. The left atrial surgical margin can be analyzed by frozen section: in cases of microscopically invaded section, the interatrial groove can be dissected a step forward to achieve a complete resection with no need for CPB.
Discussion
Lung cancers invading the heart are only a small subset of T4 tumors. Acquiring sufficient randomized data in order to provide evidence-based medicine will likely never be possible. Therefore, we must analyze reported experiences to make clinical decisions.
We should first define what is considered to be an invasion of the left atrium: we believe the cornerstone of the definition is the presence of cancer cells within cardiac muscle fibers on the pathologic specimen; it means that intrapericardial section of the pulmonary veins should not be classified as extended resection to the left atrium (involving a down-staging from T4 to T3 tumors). This precise point is almost never assessed in the surgical literature.
The impact of neoadjuvant therapy is still debated (6-8) especially in the subset of cT4N2 disease. We believe the aim of induction chemotherapy or chemoradiotherapy is to downstage locally advance NSCLC by reducing tumor size and lymph node involvement, as well as potentially eradicating micrometastatic disease. To date, very little data are available and no recommendation can be make. Most of the reported studies involving NSCLC invading the left atrium did not assess this issue.
Here are summarized the main results of reported retrospective studies involving pulmonary resection extended to the left atrium (Table 1).

Full table
Unanimously, good survival rates were obtained by resection of T4 tumors in cases of N0 or N1 nodal status and if complete surgical resection is achieved. The influence of the N status on survival is well recognized (15). Fukuse and colleagues (9) reported a median survival time of 29 months in patients T4N0 NSCLC and 9 months in T4N2 NSCLC. It emphasizes the need for accurate mediastinal staging to exclude patients with occult N2 disease who would have only few benefits from aggressive surgical therapy.
It is also clear that operative morbidity and mortality of extended resection have improved over time, yet the risks are still high. Such procedures should only be suitable for highly selected patients: i.e., those with a minor medical history, and who have good physical health and nutritional status.
We achieved a 5-year survival rate of 43.7%. This result is higher than that reported in the literature, which ranges from 0-36%. This difference may be due to several factors including the rate of complete R0 resection ranging from 35-100%.
A large majority (all postoperative survivors but one) of patients (68.4%) underwent complete neoadjuvant therapy, and 84.2% also underwent complete adjuvant therapy including at least 50 grays of radiation therapy and chemotherapy.
Although pathologic N2 status is considered a predictor for a poor prognosis (16), in our study, pN2 status did not appear to be a pejorative risk factor, probably because of the smaller number of patients. Persistent N2 after induction therapy probably don’t benefit from surgery, confirming the need for accurate restaging. Mediastinoscopy and/or EUS-EBUS-TBNA should be of systematic use to evaluate this therapeutic response.
We reported (5) an overall postoperative morbidity rate of 52.6% (n=10). The postoperative mortality rate was 10.5% (n=2) and the 90-day mortality rate was 15.7% (n=3). The 5-year survival rate was 43.7%, and the median survival time was 28.9 months. The recurrence rate was 21.0%. Three patients (15.7%) were alive for >6 years post-surgery and were considered cured.
Conclusions
Extended lung surgery with partial resection of the left atrium is a feasible procedure with acceptable morbidity and mortality rates. In highly selected patients, who eventually underwent neoadjuvant treatment, it enabled complete resection of the disease. Current data suggest that resection should be considered in such advanced tumors in relation to multidisciplinary care. Probably, experienced, high volume centers are best suitable to offer such therapeutic options. This resulted in improved long-term survival, which should be the surgeon’s goal.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Goldstraw P, Crowley J, Chansky K, et al. The IASLC Lung Cancer Staging Project: proposals for the revision of the TNM stage groupings in the forthcoming (seventh) edition of the TNM Classification of malignant tumours. J Thorac Oncol 2007;2:706-14. [PubMed]
- Bailey CP, Schechter DC, Folk FS. Extending operability in lung cancer involving the heart and great vessels. Ann Thorac Surg 1971;11:140-50. [PubMed]
- Muralidaran A, Detterbeck FC, Boffa DJ, et al. Long-term survival after lung resection for non-small cell lung cancer with circulatory bypass: a systematic review. J Thorac Cardiovasc Surg 2011;142:1137-42. [PubMed]
- Yamamoto N. Experimental study of combined left atrium resection for lung cancer. Nihon Kyobu Geka Gakkai Zasshi 1986;34:958-65. [PubMed]
- Galvaing G, Tardy MM, Cassagnes L, et al. Left atrial resection for T4 lung cancer without cardiopulmonary bypass: technical aspects and outcomes. Ann Thorac Surg 2014;97:1708-13. [PubMed]
- Martins RG, D’Amico TA, Loo BW Jr, et al. The management of patients with stage IIIA non-small cell lung cancer with N2 mediastinal node involvement. J Natl Compr Canc Netw 2012;10:599-613. [PubMed]
- Albain KS, Swann RS, Rusch VW, et al. Radiotherapy plus chemotherapy with or without surgical resection for stage III non-small-cell lung cancer: a phase III randomised controlled trial. Lancet 2009;374:379-86. [PubMed]
- van Meerbeeck JP, Kramer GW, Van Schil PE, et al. Randomized controlled trial of resection versus radiotherapy after induction chemotherapy in stage IIIA-N2 non-small-cell lung cancer. J Natl Cancer Inst 2007;99:442-50. [PubMed]
- Fukuse T, Wada H, Hitomi S. Extended operation for non-small cell lung cancer invading great vessels and left atrium. Eur J Cardiothorac Surg 1997;11:664-9. [PubMed]
- Ratto GB, Costa R, Vassallo G, et al. Twelve-year experience with left atrial resection in the treatment of non-small cell lung cancer. Ann Thorac Surg 2004;78:234-7. [PubMed]
- Bobbio A, Carbognani P, Grapeggia M, et al. Surgical outcome of combined pulmonary and atrial resection for lung cancer. Thorac Cardiovasc Surg 2004;52:180-2. [PubMed]
- Spaggiari L, D’Aiuto M, Veronesi G, et al. Extended pneumonectomy with partial resection of the left atrium, without cardiopulmonary bypass, for lung cancer. Ann Thorac Surg 2005;79:234-40. [PubMed]
- Wu L, Xu Z, Zhao X, et al. Surgical treatment of lung cancer invading the left atrium or base of the pulmonary vein. World J Surg 2009;33:492-6. [PubMed]
- Wang XX, Liu TL, Yin XR. Surgical treatment of IIIb-T4 lung cancer invading left atrium and great vessels. Chin Med J (Engl) 2010;123:265-8. [PubMed]
- Bernard A, Bouchot O, Hagry O, et al. Risk analysis and long-term survival in patients undergoing resection of T4 lung cancer. Eur J Cardiothorac Surg 2001;20:344-9. [PubMed]
- Vansteenkiste JF, De Leyn PR, Deneffe GJ, et al. Survival and prognostic factors in resected N2 non-small cell lung cancer: a study of 140 cases. Leuven Lung Cancer Group. Ann Thorac Surg 1997;63:1441-50. [PubMed]



