Radiotherapy in early stage classical Hodgkin lymphoma: past, present and future
In 1832, Dr. Thomas Hodgkin first described Hodgkin’s disease, and opened a long road to understand and research on malignant lymphoma. Hodgkin lymphoma (HL) is a subtype of malignant lymphoma, and which in west countries is more common than in China. The subtypes of nodular sclerosis and mixed cellularity in HL are same common in China, however the nodular sclerosis is the most subtype in west countries. Extended field radiotherapy (EFRT) alone had significantly improved local control rate and survival of lymphoma patients than ever, especially for early stage patients. Now the main treatment modality is combined modality therapy (CMT), including chemotherapy (CT) and radiotherapy (RT). RT is still an important treatment to decrease local recurrence and prolong survival, but the trend is smaller radiation field and lower dose in combined treatment modality. CT alone has been tried in some clinical trials, but no clear conclusion. The target drugs, such as some monoclonal antibodies, have improved survival of patients with HL, which may be affect the treatment modality for this disease.
Comparing involved field radiotherapy (IFRT) and EFRT for early stage of HL
In the past 40 years, some significant changes in the management of HL have been occurred. The use of high-dose (over 40 Gy) EFRT alone in patients with stage I or II HL showed 85% to 90% in 10-year survival (1). However, late complications of irradiation, including second neoplasms, heart infarction, myelodysplastic syndromes (MDS), hypothyroidism and so on, which are not unique in patients treated for HL, and counteract the efficacy of these treatments (2-5). The actuarial risk of second malignancies following treatment for HL that is most attributable to RT has been reported to be 21-27% at 25-30 years (6-8). An international retrospective study by the National Cancer Institute of 32,591 HL patients demonstrated a 25-year actuarial risk of solid malignancy of 21.9% (8). Superior results have been obtained with CMT, which means the IFRT following CT. When tumor remission after CT, patient receives IFRT, of which volume of RT is much smaller than EFRT. This treatment modality has been considered as the standard therapy for early stage HL. Franklin et al. reported a meta-analysis review of 23-group randomized controlled trials, including 3,888 cases with early stage HL. The results of 13 groups randomized studies showed that groups in EFRT and in CMT, 10-year recurrence rates were 32.7% and 15.8%, respectively (P<0.0001), 10-year OS were 76.5% and 79.4%, respectively (P>0.05). The conclusion was that combined therapy improved DFS, but no difference in OS. The second malignancy risks (SMR) were lower with CMT than with EFRT alone as initial treatment (P=0.03). No significant difference in SMR between IFRT and EFRT (P=0.28), but more breast cancers occurred with EFRT (P=0.04). SMR were marginally higher with CMT than with CT alone as initial treatment (P=0.05) (9). Therefore, doctors have been trying to decrease the volume of RT following an appropriate CT.
RT for favorable early stage HL
For favorable early stage HL, a number of prospective randomized studies have established combined modality treatment as the standard therapy (10-12). Fermé et al. reported the H8-F study on favorable early stage HL. Totally 542 patients were eligible for H8-F trial and underwent randomization into two arms. The study compared MOPP-ABV ×3 plus IFRT with subtotal nodal irradiation alone (STNI, reference group). The median follow-up was 92 months. In the H8-F trial, the estimated 5-year EFS was significantly higher after MOPP-ABV ×3 plus IFRT than after STNI alone (98% vs. 74%, P<0.001). The 10-year OS estimates were 97% and 92%, respectively (P=0.001) (10). Bonadonna et al. [2004] reported a study on early stage HL. A total of 136 eligible patients were entered onto this trial. According the randomization after CT, in 66 patients RT consisted of subtotal nodal plus spleen irradiation, whereas in 70 patients RT was limited to the previous involved sites only (IFRT). Complete remission was achieved in 100% and in 97% of patients in two arms, respectively. The 12-year freedom from progression rates were 93% after ABVD and STNI, and 94% after ABVD and IFRT, whereas the overall survivals were 96% and 94%, respectively. There are no differences in FFP and OS between ABVD-STNI arm and ABVD-IFRT arm (11). The results of above trials show that IFRT is sufficient treatment after a CT-induced complete remission has been obtained and that EFRT or STNI whether alone or after CT can no longer be recommended.
Some studies have focused on more reducing the CT and/or limiting the RT. The GHSG HD-7 study compared ABVD ×2 plus EFRT 30 Gy (IFRT boost 10 Gy) to EFRT 30 Gy alone (IFRT boost 10 Gy) in favorable stages I and II patients (13). The freedom from treatment failure (FFTF) was 96% for combine modality treatment, whereas 84% for EFRT alone. However, the OS of both groups were 94%. Therefore, the reduction is possible, not only in the size of the RT fields, but also in the cycle number of CT. The GHSG reported the HL10 trial result in 2010 (14). The large trial comprised of 1,190 patients with favorable clinical stages I and II were randomized to ABVD ×4 plus 30 Gy IFRT, ABVD ×4 plus 20 Gy IFRT, ABVD ×2 plus 30 Gy IFRT or ABVD ×2 plus 20 Gy IFRT. The results showed that there is no difference between 2 and 4 cycles ABVD in survival; moreover, no difference between IFRT 20 and 30 Gy survivals after CT (Table 1). Therefore, 2 cycles of ABVD + IFRT 20-30 Gy as the standard regimen is sufficiently effective and safe for patients with favorable early stage HL.

Full table
RT for unfavorable early stage HL
It is strongly recommended that patients in the unfavorable early stage of HL should receive a combination of CT followed by IFRT with a total dose of 30 Gy. There are some randomized studies to compare EFRT and IFRT following CT. In GHSG HD8 trial, 1,204 patients with unfavorable clinical stages I and II were randomized to group A (COPP/ABVD ×4 + EFRT 30 Gy + Bulk 10 Gy) and group B (COPP/ABVD ×4 + IFRT 30Gy + Bulk 10 Gy). The 5-year FFTF were 86% and 84% respectively, the 5-year OS were 91% and 92%, respectively. The result showed that there is no significant difference between group A (EFRT) and group B (IFRT) in FFTF and OS (15). In H8-U trial, it is similar result of survivals between STNI and IFRT following CT (10). The GHSG HL11 [1998-2003] trial randomized of unfavorable patients with CS I and II to ABVD ×4 + IFRT (30 Gy), ABVD ×4 + IFRT (20 Gy), BEACOPP baseline + IFRT (30 Gy), BEACOPP baseline + IFRT (20 Gy). Four cycles of ABVD + IFRT (30 Gy) was no difference to BEACOPP baseline + IFRT (20 or 30 Gy) in survivals, and the toxicities in ABVD were milder than BEACOPP baseline. However, the group with ABVD ×4 + IFRT (20 Gy) was inferior to other groups in survival (Table 2) (16). Therefore, the regimen of ABVD ×4 + IFRT (30 Gy) was considered as standard treatment. In HD14 trial, 2+2 CT (two cycles BEACOPPesc followed by two cycles of ABVD) can improve PFS of patients with four cycles ABVD, but it cannot improve OS of patients, and with more severe toxicities (17). Therefore, involved field irradiation (30 Gy) following CT (ABVD ×4) is recommended for unfavorable early stage patients.
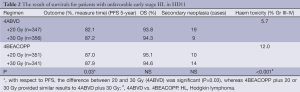
Full table
Involved node radiotherapy (INRT): a smaller volume of RT
The INRT has shown benefit in critical organ irradiation in limited stage HL. This method, first developed by EORTC/GELA group, has less harmful for healthy tissues and further reduction in SMR. The INRT is designed to eradicate involved lymph nodes, while the IFRT focus on involved lymph node region, which volume of RT may be enlarged after CT (18). Campbell et al. compared with IFRT, INRT signifiantly reduced mean doses to lungs (P<0.01), breasts (P<0.01), thyroid (P<0.01) and heart (P<0.01) (19). And the relapse rate of HL in INRT (3%) was similar to in IFRT (5%) and in EFRT (3%) (20). Maraldo et al. reported a retrospective analysis using INRT (30-36 Gy) following CT (ABVD) to treat 97 clinical stage I-II HL patients. The 4-year PFS and OS were 96.4% and 94%, respectively. Early toxicity of radiation therapy was limited to grade 1 (23.4%) and grade 2 (13.8%). During follow-up, 7 secondary malignancies were diagnosed, and 5 patients developed heart disease. INRT offers excellent tumor control without more severe toxicity than IFRT or EFRT (21). Pre-CT FDG-PET leads to significantly better INRT delineation without necessarily increasing radiation volumes (22). In future, it is necessary to have some randomized studies on INRT for making it into as a standard method of treatment.
Whether or not the RT can been omitted?
According to a series of clinical studies, the volume and dosage of RT have been reduced significantly in early stage HL. Whether the RT can been omitted? A randomized study (NCI-C/ECOG HD6 trial and 399 cases) on non-bulky early HL compared 4-6 cycles of ABVD alone with STNI with or without 2 cycles ABVD for favorable patients or for unfavorable patients. The result showed the over survival in ABVD alone was higher than in STNI (94% vs. 87%, P=0.04), but the freedom from disease progression was lower (87% vs. 92%, P=0.05), especially in unfavorable patients group (86% vs. 94%, P=0.006). The mortality in STNI group was higher than in ABVD alone group mainly due to late treatment complications such as second cancers, related infections, cardiac events and so on. Therefore, the patients in STNI group had lower recurrence rate and higher treatment complication rate than in ABVD alone, especially in unfavorable cases. These complications decreased OS in STNI group. So that further study on comparing IFRT or INRT with ABVD alone should be considered (23).
Some randomized clinical trials have shown that CT alone lead to a higher recurrence rate and shorter PFS. EORTC-GELA H9F compared 6 cycles of EBVP with or without 20 or 36 Gy IFRT. There was an excessive number of relapses occurring in an EBVP alone arm, so that the EORTC had to prematurely close their H9F trial (24). In RAPID study, 602 patients with negative interim PET after ABVD ×3 CT were further randomized into two arms: IF-RT or observation arm. The 3-year PFS in group of IF-RT and group of observation were 97% and 90.7%, respectively (P=0.03). Patients with positive interim PET received both one ABVD cycle and IF-RT, and their 3-year PFS and OS were 85.9% and 93.9% respectively (25). EORTC H10 study had a similar result in interim analysis (26). Therefore, the RT following CT still is standard therapy for early stage HL. Some clinical trials on PET guiding are ongoing, such as GHSG HD16 and HD17.
The outlook
At present, great changes have occurred in radiation therapy technology. With the development of computer technology, RT treatment planning systems are more perfect. The techniques of Intensity-Modulated Radiation Therapy (IMRT) and Image Guided Radiation Therapy (IGRT) makes RT dose distribution more precisely conform to the target volume from small and regular tumor to large and irregular tumor. Proton accelerators have been applied in clinical treatment, which have significant advantages in radiation dose distribution in tumor target volume. In recent years, the emergence of IMRT, gamma knife, VAMT, Tomotherapy, Cyber-knives, 4D-techenique and other new technological equipment, significantly improve the radiation dose distribution in the tumor target volume, and significantly decrease the radiation dose in the normal tissues and organs (19,27,28). According to these new techniques, at the same time of controlling tumor, we can protect the normal tissues and organs, improve the quality of life, reduce damage of tissues and organs function, and also reduce the incidence of second cancers. Although some of above techniques has mainly used in other cancers, such as lung cancer, liver cancer, and so on. PET/CT displays the sites of living tumor tissue is superior to CT and other imaging techniques, so that it is effecting on the RT whether or not to do and how to do, which has been widely used in clinical practice (29-32). In future, there will be new different indicators of PET/CT for different purposes of diagnosis and treatment, or some other new techniques, which can more accurately and sensitively shows the active part of tumor. These technologies will make the design and realizing this design on tumor target volume of RT more accurately.
Conclusions
According to a series of studies for many years, as the first-line therapy on patients with early-stage HL, combined therapy has replaced the RT alone. The recommended regimens are 2 cycles of ABVD plus IFRT (20-30 Gy) for the favorable patients, and 4 cycles of ABVD plus IFRT (30 Gy) for unfavorable patients. The INRT has shown the potential to achieve satisfactory primary tumor control with lower treatment-related toxicity than EFRT or IFRT in combined therapy. Some prospective randomized studies are going about CT plus INRT under the guidance of PET. It is not certain that whether CT alone is more beneficial or not to cure limited HL. Appropriate application of new RT techniques can improve the radiation dose distribution in target fields and protect normal tissues from excess RT-related damage.
Acknowledgements
Disclosure: The author declares no conflict of interest.
References
- Mauch PM, Weinstein H, Botnick L, et al. An evaluation of long-term survival and treatment complications in children with Hodgkin’s disease. Cancer 1983;51:925-32. [PubMed]
- Mariotto AB, Rowland JH, Yabroff KR, et al. Long-term survivors of childhood cancers in the United States. Cancer Epidemiol Biomarkers Prev 2009;18:1033-40. [PubMed]
- Castellino SM, Geiger AM, Mertens AC, et al. Morbidity and mortality in long-term survivors of Hodgkin lymphoma: a report from the Childhood Cancer Survivor Study. Blood 2011;117:1806-16. [PubMed]
- Hodgson DC, Hudson MM, Constine LS. Pediatric hodgkin lymphoma: maximizing efficacy and minimizing toxicity. Semin Radiat Oncol 2007;17:230-42. [PubMed]
- Cella Laura, Conson Manuel, Pressello Maria Cristina, et al. Hodgkin’s lymphoma emerging radiation treatment techniques: trade-offs between late radio-induced toxicities and secondary malignant neoplasms. Radiation Oncology 2013;8:22. [PubMed]
- Ibrahim EM, Kazkaz GA, Abouelkhair KM, et al. Increased risk of second lung cancer in Hodgkin’s lymphoma survivors: a meta-analysis. Lung 2013;191:117-34. [PubMed]
- Ibrahim EM, Abouelkhair KM, Kazkaz GA, et al. Risk of second breast cancer in female Hodgkin’s lymphoma survivors: a meta-analysis. Cancer 2012;12:197. [PubMed]
- Dores GM, Metayer C, Curtis RE, et al. Second malignant neoplasms among long-term survivors of Hodgkin’s disease: a population-based evaluation over 25 years. J Clin Oncol 2002;20:3484-94. [PubMed]
- Franklin J, Pluetschow A, Paus M, et al. Second malignancy risk associated with treatment of Hodgkin’s lymphoma: meta-analysis of the randomised trials. Ann Oncol 2006;17:1749-60. [PubMed]
- Fermé C, Eghbali H, Meerwaldt JH, et al. Chemotherapy plus involved-field radiation in early-stage Hodgkin’s disease. N Engl J Med 2007;357:1916-27. [PubMed]
- Bonadonna G, Bonfante V, Viviani S, et al. ABVD plus subtotal nodal versus involved-field radiotherapy in early-stage Hodgkin’s disease: long-term results. J Clin Oncol 2004;22:2835-41. [PubMed]
- Press OW, LeBlanc M, Lichter AS, et al. Phase III randomized intergroup trial of subtotal lymphoid irradiation versus doxorubicin, vinblastine, and subtotal lymphoid irradiation for stage IA to IIA Hodgkin’s disease. J Clin Oncol 2001;19:4238-44. [PubMed]
- Wiedenmann S, Schiller P, Paulus U, et al. Treatment of early and intermediate stage Hodgkin’s lymphoma in the German Hodgkin’s Lymphoma Study Group. Ann Oncol 2002;13:84-5. [PubMed]
- Engert A, Plütschow A, Eich HT, et al. Reduced treatment intensity in patients with early-stage Hodgkin’s lymphoma. N Engl J Med 2010;363:640-52. [PubMed]
- Engert A, Schiller P, Josting A, et al. Involved-field radiotherapy is equally effective and less toxic compared with extended-field radiotherapy after four cycles of chemotherapy in patients with early-stage unfavorable Hodgkin’s lymphoma: results of the HD8 trial of the German Hodgkin’s Lymphoma Study Group. J Clin Oncol 2003;21:3601-8. [PubMed]
- Eich HT, Diehl V, Görgen H, et al. Intensified chemotherapy and dose-reduced involved-field radiotherapy in patients with early unfavorable Hodgkin’s lymphoma: final analysis of the German Hodgkin Study Group HD11 trial. J Clin Oncol 2010;28:4199-206. [PubMed]
- von Tresckow B, Plütschow A, Fuchs M, et al. Dose-intensification in early unfavorable Hodgkin’s lymphoma: final analysis of the German Hodgkin Study Group HD14 trial. J Clin Oncol 2012;30:907-13. [PubMed]
- Weber DC, Peguret N, Dipasquale G, et al. Involved-node and involved-field volumetric modulated arc vs. fixed beam intensity-modulated radiotherapy for female patients with early-stage supra-diaphragmatic Hodgkin lymphoma: a comparative planning study. Int J Radiat Oncol Biol Phys 2009;75:1578-86. [PubMed]
- Campbell BA, Hornby C, Cunninghame J, et al. Minimising critical organ irradiation in limited stage Hodgkin lymphoma: a dosimetric study of the benefit of involved node radiotherapy. Ann Oncol 2012;23:1259-66. [PubMed]
- Campbell BA, Voss N, Pickles T, et al. Involved-nodal radiation therapy as a component of combination therapy for limited-stage Hodgkin’s lymphoma: a question of field size. J Clin Oncol 2008;26:5170-4. [PubMed]
- Maraldo MV, Aznar MC, Vogelius IR, et al. Involved node radiation therapy: an effective alternative in early-stage hodgkin lymphoma. Int J Radiat Oncol Biol Phys 2013;85:1057-65. [PubMed]
- Girinsky T, Aupérin A, Ribrag V, et al. Role of FDG-PET in the implementation of involved-node radiation therapy for Hodgkin lymphoma patients. Int J Radiat Oncol Biol Phys 2014;89:1047-52. [PubMed]
- Meyer RM, Gospodarowicz MK, Connors JM, et al. ABVD alone versus radiation-based therapy in limited-stage Hodgkin’s lymphoma. N Engl J Med 2012;366:399-408. [PubMed]
- Thomas J, Fermé C, Noordijk EM, et al. Results of the EORTC-GELA H9 randomized trials: the H9-F trial (comparing 3 radiation dose levels) and H9-U trial (comparing 3 chemotherapy schemes) in patients with favorable or unfavorable early stage Hodgkin’s lymphoma (HL). Haematologica 2007;92:27. [PubMed]
- Radford J, Barrington S, Counsell N, et al. Involved Field Radiotherapy Versus No Further Treatment in Patients with Clinical Stages IA and IIA Hodgkin Lymphoma and a ‘Negative’ PET Scan After 3 Cycles ABVD. Results of the UK NCRI RAPID Trial. Blood (ASH Annual Meeting Abstracts) 2012;120:abstr 547.
- Raemaekers JM, André MP, Federico M, et al. Omitting radiotherapy in early positron emission tomography-negative stage I/II Hodgkin lymphoma is associated with an increased risk of early relapse: Clinical results of the preplanned interim analysis of the randomized EORTC/LYSA/FIL H10 trial. J Clin Oncol 2014;32:1188-94. [PubMed]
- Matoba M, Oota K, Toyoda I, et al. Usefulness of 4D-CT for radiation treatment planning of gastric MZBCL/MALT. J Radiat Res 2012;53:333-7. [PubMed]
- Moorrees J, Bezak E. Four dimensional radiotherapy: a review of current technologies and modalities. Australas Phys Eng Sci Med 2012;35:399-406. [PubMed]
- Somer EJ, Pike LC, Marsden PK. Recommendations for the use of PET and PET-CT for radiotherapy planning in research projects. Br J Radiol 2012;85:e544-8. [PubMed]
- Yeoh KW, Mikhaeel NG. Are we ready for positron emission tomography/computed tomography-based target volume definition in lymphoma radiation therapy? Int J Radiat Oncol Biol Phys 2013;85:14-20. [PubMed]
- Sehn LH, Hoskins P, Klasa P, et al. FDG-PET Scan Guided Consolidative Radiation Therapy Optimizes Outcome In Patients with Advanced-Stage Diffuse Large B-Cell Lymphoma (DLBCL) with Residual Abnormalities on CT Scan Following R-CHOP. Blood (ASH Annual Meeting Abstracts) 2010;116:abstr 854.
- Aridgides P, Bogart J, Shapiro A, et al. PET Response-Guided Treatment of Hodgkin’s Lymphoma: A Review of the Evidence and Active Clinical Trials. Adv Hematol 2011;2011:309237.


