The changing world of oncology drug development—A global pharmaceutical company’s perspective
Over the last couple of decades compared to the 1990s the success rates for drug development have fallen significantly across all therapeutic areas, including oncology and this decline has been widely discussed with some articles implying further inevitable decline (1,2). However looking in more recent years there is clear evidence of innovative new medicines making it successfully through the pharmaceutical industry pipelines. Since 2011, 27 novel cancer medicines have had a first indication approved by the US FDA (Table 1). The pattern emerging from the successfully developed drugs is that an increasing proportion are targeted to patient populations where cancer growth is driven by a pathway specifically inhibited by the drug. The emerging deeper understanding of molecular drivers of cancer through technology improvements and cost reductions in next generation sequencing has presented opportunities to create new approvable development paths for novel therapeutic agents. Another exciting emerging trend is the understanding of the mechanisms or ‘checkpoints’ controlling the development of an immunosuppressive environment within tumors and the development of novel medicines which can affect these ‘checkpoints’ and induce prolonged responses based on reactivation of immune-driven tumor cell killing (3,4). However, these same opportunities also present challenges which require adaptation of the ‘traditional’ drug development route starting from phase I. Success is indeed possible, however it requires good understanding of the driving biology, the right preclinical models, understanding of the limitations of these models, and close collaboration between drug developers, diagnostic developers, academic investigators and regulatory bodies.
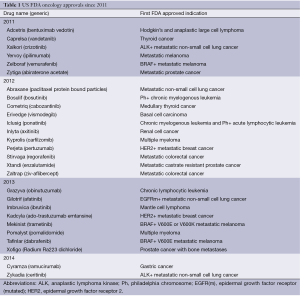
Full table
Better understanding of the driving biology in cancer has come from the efforts to document the genetic aberrations underpinning the development and progression of cancers—efforts such as The Cancer Genome Atlas project (http://cancergenome.nih.gov/) and the International Cancer Genome Consortium (http://www.icgc.org/) to create a comprehensive catalogue of significantly mutated genes across all major cancer types (5-7). The output from such large scale projects is radically transforming the way cancer science is conducted. At the same time, these large-scale efforts have demonstrated an extremely high level of genome complexity therefore highlighting the need for high-throughput technologies to assess the functional importance of the aberrations identified by these analyses. One of the major challenges is determining which aberrations are ‘drivers’ of tumor growth versus contributors versus passengers (8,9). If we are to increase the probability of success in drug development this will only come via a thorough mechanistic understanding of a target’s cancer activity and the specific biological and genotypic context in which it operates. Another challenge even when a genetic aberration has successfully been validated as a driving oncogenic change is to understand tumor heterogeneity (10). Is the genetic aberration present with a high allelic frequency throughout the tumor or only present in a dominant clone or subclone of the tumor cell population? A subclonal genetic event may be important for the emergence of resistance, but clearly monotherapy with a medicine directed only to a subclonal genetic event is unlikely to lead to a high or durable response rate.
It is instructive to examine how this emerging knowledge has affected the approach to drug development since the time when gefitinib was taken through phase I to III development, compared with the current development of novel targeted therapies in lung cancer. Gefitinib entered phase I clinical trials in 1998 (11,12). In those early clinical trials evidence of major tumor regression was seen in a small proportion of patients with non-small cell lung cancer (NSCLC)—such responses were seen across the dose range 150 to 800 mg/day, while the majority of dose interruptions and reductions due to toxicity were required in patients receiving more than 600 mg/day. From these data, two doses (250 and 500 mg/day) were selected for investigation in phase II and phase III trials in an unselected patient population in NSCLC (13-15). The phase II IDEAL trial in which around 50% of the enrolled patients were Japanese demonstrated an encouraging response rate of 18-19% (13). However, the phase III trials adding gefitinib onto standard of care chemotherapy subsequently failed to show an improvement in overall survival or substantive improvement in progression free survival (PFS) in the unselected patient population (14,15). Only later in 2004 was the discovery made that the ‘super-responders’ to gefitinib had a mutation in the epidermal growth factor receptor (EGFR) ATP binding pocket which was shown to be a tumor ‘driver’ leading to addiction of the tumor to signaling through this pathway (16,17). Inhibition of that driving signal by treatment with gefitinib leads not just to inhibition of proliferation, but to apoptosis of the addicted cell population (18). These mutations are more common in patients with adenocarcinoma, females, Asian patients, and those who do not have a smoking history (19). When patients are selected based on the presence of a sensitizing mutation in EGFR, the response rates to gefitinib or another EGFR inhibitor erlotinib are >60% with median PFS of 9 to 12 months (20). The IPASS trial, conducted in East Asian sites which randomized patients to either gefitinib or standard of care chemotherapy and enriched for patients likely to have EGFR mutations by including female light or never-smokers, demonstrated a statistically significant improvement in PFS in the sub-group with EGFR mutations (hazard ratio 0.48, P<0.001) but was associated with a significantly shorter PFS among patients with wild-type EGFR (21). Gefitinib was initially approved in Japan in 2002, the first EGFR-tyrosine kinase (TK) inhibitor to be approved for use in lung cancer. It was also approved by the US FDA in 2003 as an accelerated approval based on durable responses in third line patients with NSCLC, but when the phase III trials failed to show a survival benefit in combination with chemotherapy or as monotherapy, the FDA modified the label for gefitinib in 2005 to limit the indication to cancer patients who, in the opinion of their treating physicians, are currently benefiting, or have previously benefited, from gefitinib treatment. In 2009 the European Commission granted marketing authorization for gefitinib for the treatment of adults with locally advanced or metastatic NSCLC with activating mutations of EGFR-TK across all lines of therapy, based on the data from IPASS and the INTEREST trials—the first drug for lung cancer to be associated with a companion diagnostic test (22).
Fast forward now to March 2013, when a candidate drug developed at AstraZeneca, AZD9291 entered clinical trials. This drug was designed to overcome a common resistance mechanism which develops in patients with a ‘driving’ EGFR mutation and treated with an EGFR inhibitor such as gefitinib or erlotinib. It is now known through genetic analysis of biopsies taken from patients who have progressed after these agents that around 60% have developed resistance because their tumors have developed a second mutation in EGFR—the so called ‘gatekeeper’ mutation T790M (23,24). This changes the binding affinity for ATP, and therefore gefitinib is displaced from the binding pocket, and the ‘driving’ signal for proliferation is switched on again.
This detailed understanding of the mechanism underlying disease progression alongside the substantial years of experience gained in kinase chemistry underpinned the ability of the discovery scientists at AstraZeneca to select AZD9291 as a candidate drug for the treatment of patients with NSCLC and a sensitizing mutation in EGFR who have developed resistance to EGFR inhibitors (25,26). The development of this drug was also driven by further understanding of the limitations of drugs such as gefitinib due to their similar potency versus the wild type EGFR compared with the mutated form. This leads to diarrhea and rash as dose-limiting toxicities. The selection of AZD9291 was based on a large margin of potency for the double mutant EGFR versus the wild type EGFR (27). The rationale was that improved efficacy would be seen if increased target inhibition could be achieved at doses well below those that would inhibit wild type EGFR and produce rash and diarrhea. Preclinical development was done using tumor xenografts with single and double EGFR mutations as well as in genetically engineered mouse models with these mutations (27). So in contrast to gefitinib’s development the relevant patient population most likely to respond to this therapy was identified well before starting phase I development and those patients were enrolled into the first clinical trials. In fact, durable clinical responses to AZD9291 were seen in the first cohort of patients treated. Data presented at the World Conference on Lung Cancer in Sydney in October 2013 demonstrated unconfirmed and confirmed responses in 9 of 18 patients with T790M mutations (28). Accrual to the phase I trial has been very rapid, with sites open in EU countries, US and in Asia. The rapid generation of data demonstrating a high and durable response rate has led to Breakthrough Therapy designation by the US FDA, and rapid entry into pivotal phase II,III trials. The recent rapid approvals of crizotinib and ceritinib in patients with NSCLC with ALK fusions are further examples of this new paradigm of very rapid drug development enabled by understanding of driving genetic aberrations, selection of the right patient population through diagnostic testing, high and durable response rates in early clinical trials. The probability of success for approval after these kinds of results in phase I is substantially higher than the industry benchmarks for programs at the start of phase III trials.
These successes are encouraging, but they really represent one step forward, rather than a complete solution to transforming the outcomes for patients with lung cancer. As the discussion above illustrated, response rates to such novel therapies can be high, but resistance inevitably emerges. The median number of mutations present in a patient’s tumor with metastatic NSCLC is around ten mutations per megabase—one of the most highly mutated cancers (29). The selection pressure generated by treatment with a targeted monotherapy leads to outgrowth of clones which have mutations with resistance mechanisms (30). The history of cancer drug development teaches us that combination therapies are required to substantially increase long term survival. We are already investing in understanding the mechanisms which might lead to resistance to treatment with AZD9291 (31), and clinical trials examining potential combinations of drugs which might overcome these resistance mechanisms will start later this year.
Another exciting development in oncology is coming from the biological insights around mechanisms controlling the immune response to cancer. The data from clinical trials with drugs such as ipilimumab an anti-CTLA4 antibody in metastatic melanoma, which inhibits a key negative regulator of T cell activation has shown that a subset of patients treated with this drug have highly durable responses measured in years rather than months (32). Similar patterns of highly durable clinical responses have also been seen with a number of agents targeting other immune ‘checkpoints’ of PD1 and the ligand PDL1, including in patients with advanced lung cancer (4). To date monotherapy response rates with these agents do not reach the same levels as targeted therapies such as gefitinib, but emerging data on potential tests which could be used to select patients suggests that such selection could increase the response rate. Another wave of clinical trials is now starting to examine the combinations of such agents with standard of care therapies, with other immunotherapies and with targeted agents. There is much to learn about the optimal way to combine these agents but the hope is that such combinations could improve both the initial response rates and the long term survival of patients with metastatic cancer.
The increased segmentation of diseases such as NSCLC, particularly beyond the relatively large groups of EGFR and Kras mutation however presents challenges to recruit patients into early phase clinical trials particularly if those segments represent less than 10% of the NSCLC population. The costs and time for screening large numbers of patients to find those eligible for such trials are considerable and such a system does not serve patients, investigators or pharmaceutical sponsors well. An emerging trend in response to this challenge is the development of ‘basket’ studies which include one or more screening tests for multiple genetic aberrations and the direction of patients to one of several arms of a clinical trial based on the specific aberration in their tumor. One such trial is being run by Cancer Research UK in collaboration with AstraZeneca and Pfizer which will provide access to up to 14 different drugs—12 from AstraZeneca and 2 from Pfizer. A second example, also in lung cancer, but in squamous lung cancer is being run by Friends of Cancer Research in the US. A third example is a collaboration AstraZeneca has with Korea’s Samsung Medical Center (SMC) for a multi-drug clinical trial. It offers gastric cancer patients the opportunity to receive a novel targeted therapy based on the molecular profile of their disease. The successful running of such trials requires both an infrastructure to support routine genetic diagnostic testing and analysis, and access to a wide enough range of experimental therapies which may benefit patients whose tumors have a specific genetic aberration. This requires novel mechanisms of collaboration across academia and industry—a more open approach to innovation.
“Open innovation” is a term that is used widely. But what does it mean for AstraZeneca? We are continually looking at ways to make ourselves more “porous” to the external environment; academia, industry and government departments. Earlier this year we launched our dedicated “Open Innovation” website, which is our “shop-front” for collaborations across all stages of drug discovery. The AstraZeneca Open Innovation platform (http://openinnovation.astrazeneca.com) helps us work at the crossroad of different scientific disciplines, where true creativity and innovation occurs and progress can be accelerated.
My vision is to be as inclusive as possible and to make it easy to partner and collaborate with AstraZeneca. One of the ways in which we managed to simplify and expand our external collaborations, was through the creation of what we call the “Oncology Toolbox”. Sharing compounds with investigators from academia has been accelerated, thanks to the quick and efficient way in which the Toolbox makes our compounds available for pre-clinical research. In the past, only AstraZeneca’s contracted partners had access to the Toolbox. Then, in November 2012 we opened the toolbox up to academic institutions with whom we had no established working relationships. Since then, it has proved popular with external research institutions. It has enhanced AstraZeneca’s reputation as a collaborative organization and brought new research opportunities to light. The Toolbox is bringing benefits to the company and external investigators, who have benefited from fast access to the cancer compounds and the targets they were aimed at. It gives them the chance to carry out high quality research to investigate cancer signaling pathways, evaluate novel combinations, develop novel cancer models, test new indications, discover novel biology and test hypotheses.
AstraZeneca has shared its compounds with academia in the past, but the Toolbox has expedited the process. It used to take an average of 154 days from receiving an application to shipping the material; now it takes an average of 66 days. The timeline has gone down by 57 percent, giving investigators confidence that they will have rapid access to molecules and making AstraZeneca their first stop for experimental material. The number of applications has gone up by 40 percent over the last twelve months. So what’s made the Toolbox so successful? At the heart of it all is a simplified Materials Transfer Agreement (MTA) template. This has fewer restrictions than a standard contract, which enables easier negotiation. Applicants use the template to request material via email. They submit a research proposal stating which compound they want and the hypothesis they want to test to an Oncology External Science Manager, who reviews their request. Lastly, lawyers for both parties negotiate an MTA. The Toolbox is truly global: 88 proposals have been approved since the start of 2013, from 12 different countries. The approval rate for compound requests to date is over 90 percent. In December 2012, during the initial phase of the program rollout in Asia, we introduced the program to eight centers in four countries; Singapore, Korea, Japan and China. As of today, we have received requests for 23 compounds. Of these, 17 have already been delivered or approved and one is still under review.
Our work with Hutchison MediPharma of China is another example of partnering to develop innovative medicines. We have a global licensing, co-development and commercialisation agreement for volitinib, a c-met inhibitor targeted for the treatment of cancers which have met amplification, over-expression or mutation (33). Volitinib entered phase I testing in China in June 2013. The study has two primary objectives: to evaluate safety and tolerability in advanced cancer patients in China and to determine the maximum tolerated dose of the drug candidate. The study will also evaluate volitinib’s preliminary efficacy against various tumors, including lung and gastric cancer.
In summary, the explosion of information about the genetic aberrations underlying cancer has led to several changes in the paradigm of how we develop drugs for this disease. I believe that transformation of the outcomes for patients with cancer will require detailed understanding of these genetic aberrations in each patient’s cancer at diagnosis and at progression, the development of both targeted therapies and novel immunotherapies and the ability to combine multiple of these agents together in effective and tolerable treatment regimens. The nature of clinical trials designed to address this new understanding is already changing and will change further. Far from there being a decline in innovation in pharmaceutical development, I see that we are in one of the most exciting times in cancer drug development with innovation in every aspect of how we discover and develop new therapies. I have no doubt that it is only by working together across academia and industry and sharing our scientific expertise that we stand the best chance of reaching our bold ambition of eliminating cancer as a cause of death.
Acknowledgements
Disclosure: Susan Galbraith is a full time employee and stock holder of AstraZeneca PLC.
References
- Pammolli F, Magazzini L, Riccaboni M. The productivity crisis in pharmaceutical R&D. Nat Rev Drug Discov 2011;10:428-38. [PubMed]
- Kola I, Landis J. Can the pharmaceutical industry reduce attrition rates? Nat Rev Drug Discov 2004;3:711-5. [PubMed]
- Ott PA, Hodi FS, Robert C. CTLA-4 and PD-1/PD-L1 blockade: new immunotherapeutic modalities with durable clinical benefit in melanoma patients. Clin Cancer Res 2013;19:5300-9. [PubMed]
- Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer 2012;12:252-64. [PubMed]
- Cancer Genome Atlas Research Network. Comprehensive genomic characterization defines human glioblastoma genes and core pathways. Nature 2008;455:1061-8. [PubMed]
- Cancer Genome Atlas Research Network. Integrated genomic analyses of ovarian carcinoma. Nature 2011;474:609-15. [PubMed]
- Cancer Genome Atlas Research Network. Comprehensive genomic characterization of squamous cell lung cancers. Nature 2012;489:519-25. [PubMed]
- Bozic I, Antal T, Ohtsuki H, et al. Accumulation of driver and passenger mutations during tumor progression. Proc Natl Acad Sci U S A 2010;107:18545-50. [PubMed]
- Raphael BJ, Dobson JR, Oesper L, et al. Identifying driver mutations in sequenced cancer genomes: computational approaches to enable precision medicine. Genome Med 2014;6:5. [PubMed]
- Burrell RA, McGranahan N, Bartek J, et al. The causes and consequences of genetic heterogeneity in cancer evolution. Nature 2013;501:338-45. [PubMed]
- Baselga J, Rischin D, Ranson M, et al. Phase I safety, pharmacokinetic, and pharmacodynamic trial of ZD1839, a selective oral epidermal growth factor receptor tyrosine kinase inhibitor, in patients with five selected solid tumor types. J Clin Oncol 2002;20:4292-302. [PubMed]
- Herbst RS, Maddox AM, Rothenberg ML, et al. Selective oral epidermal growth factor receptor tyrosine kinase inhibitor ZD1839 is generally well-tolerated and has activity in non-small-cell lung cancer and other solid tumors: results of a phase I trial. J Clin Oncol 2002;20:3815-25. [PubMed]
- Fukuoka M, Yano S, Giaccone G, et al. Multi-institutional randomized phase II trial of gefitinib for previously treated patients with advanced non-small-cell lung cancer (The IDEAL 1 Trial) J Clin Oncol 2003;21:2237-46. [PubMed]
- Herbst RS, Giaccone G, Schiller JH, et al. Gefitinib in combination with paclitaxel and carboplatin in advanced non-small-cell lung cancer: a phase III trial--INTACT 2. J Clin Oncol 2004;22:785-94. [PubMed]
- Giaccone G, Herbst RS, Manegold C, et al. Gefitinib in combination with gemcitabine and cisplatin in advanced non-small-cell lung cancer: a phase III trial--INTACT 1. J Clin Oncol 2004;22:777-84. [PubMed]
- Lynch TJ, Bell DW, Sordella R, et al. Activating mutations in the epidermal growth factor receptor underlying responsiveness of non-small-cell lung cancer to gefitinib. N Engl J Med 2004;350:2129-39. [PubMed]
- Paez JG, Jänne PA, Lee JC, et al. EGFR mutations in lung cancer: correlation with clinical response to gefitinib therapy. Science 2004;304:1497-500. [PubMed]
- Sordella R, Bell DW, Haber DA, et al. Gefitinib-sensitizing EGFR mutations in lung cancer activate anti-apoptotic pathways. Science 2004;305:1163-7. [PubMed]
- Sharma SV, Bell DW, Settleman J, et al. Epidermal growth factor receptor mutations in lung cancer. Nat Rev Cancer 2007;7:169-81. [PubMed]
- Soria JC, Mok TS, Cappuzzo F, et al. EGFR-mutated oncogene-addicted non-small cell lung cancer: current trends and future prospects. Cancer Treat Rev 2012;38:416-30. [PubMed]
- Mok TS, Wu YL, Thongprasert S, et al. Gefitinib or carboplatin-paclitaxel in pulmonary adenocarcinoma. N Engl J Med 2009;361:947-57. [PubMed]
- Kim ES, Hirsh V, Mok T, et al. Gefitinib versus docetaxel in previously treated non-small-cell lung cancer (INTEREST): a randomised phase III trial. Lancet 2008;372:1809-18. [PubMed]
- Kobayashi S, Boggon TJ, Dayaram T, et al. EGFR mutation and resistance of non-small-cell lung cancer to gefitinib. N Engl J Med 2005;352:786-92. [PubMed]
- Pao W, Miller VA, Politi KA, et al. Acquired resistance of lung adenocarcinomas to gefitinib or erlotinib is associated with a second mutation in the EGFR kinase domain. PLoS Med 2005;2:e73. [PubMed]
- Ward RA, Anderton MJ, Ashton S, et al. Structure- and reactivity-based development of covalent inhibitors of the activating and gatekeeper mutant forms of the epidermal growth factor receptor (EGFR). J Med Chem 2013;56:7025-48. [PubMed]
- Finlay MR, Anderton M, Ashton S, et al. Discovery of and first disclosure of the clinical candidate AZD9291, a potent and selective third-generation EGFR inhibitor of both activating and T790M resistant mutations that spares the wild type form of the receptor. [abstract]. In: Proceedings of the AACR-NCI-EORTC International Conference: Molecular Targets and Cancer Therapeutics; 2013 Oct 19-23; Boston, MA. Philadelphia (PA): AACR; Mol Cancer Ther 2013;12:Abstract nr B94.
- Cross D, Ashton S, Nebhan C, et al. AZD9291: an irreversible, potent and selective third generation tyrosine kinase inhibitor (TKI) targeting EGFR activating (EGFRm+) and resistance (T790M) mutations in advanced lung adenocarcinoma. [abstract]. In: Proceedings of the AACR-NCI-EORTC International Conference: Molecular Targets and Cancer Therapeutics; 2013 Oct 19-23; Boston, MA. Philadelphia (PA): AACR; Mol Cancer Ther 2013;12(11 Suppl):Abstract nr A109.
- Ranson M, Pao W, Kim DW, et al. AZD9291: an irreversible, potent and selective tyrosine kinase inhibitor (TKI) of activating (EGFRm+) and resistance (T790M) mutations in advanced NSCLC. World Conference on Lung Cancer Syndey Australia October 2013.
- Alexandrov LB, Nik-Zainal S, Wedge DC, et al. Signatures of mutational processes in human cancer. Nature 2013;500:415-21. [PubMed]
- Takahashi K, Kohno T, Matsumoto S, et al. Clonal and parallel evolution of primary lung cancers and their metastases revealed by molecular dissection of cancer cells. Clin Cancer Res 2007;13:111-20. [PubMed]
- Eberlein C, Ratcliffe L, O’Brien L, et al. Investigating resistance to AZD9291. Abstract in Proc American Association of Cancer Research Annual Meeting April 2014 San Diego.
- Prieto PA, Yang JC, Sherry RM, et al. CTLA-4 blockade with ipilimumab: long-term follow-up of 177 patients with metastatic melanoma. Clin Cancer Res 2012;18:2039-47. [PubMed]
- Su W, Dai G, Hong J, et al. Discovery, preclinical and early clinical evaluation of volitinib: a potent and selective c-Met kinase inhibitor. Abstract in Proc American Association of Cancer Research Annual Meeting April 2014 San Diego.


